 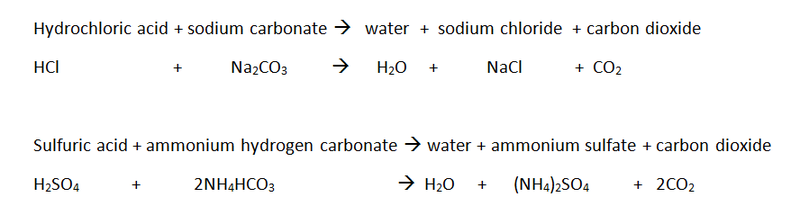
And let’s not forget that other reason why people might like fizzy drinks: they contain a lot of sugar! It’s like why some people really enjoy spicy food. 
Some scientists believe that humans like fizzy drinks because we like a bit of risk in our lives. In fact, studies have found that many other mammals actually avoid drinking carbonated liquids. Why do some people enjoy the tingling sensation of carbonated drinks in the first place? Scientists are not entirely sure. A few years later, a Swiss scientist named Jacob Schweppe created the world’s first soft drink: Schweppe’s Tonic Water! Why do people like drinking pop? The carbonation process was invented by a famous English chemist named Joseph Priestley in 1767. As the amount of H 2CO 3 in the beverage goes down, so does the soda’s ability to bring about that tingling sensation on your tongue. As that happens, the concentration of carbonic acid in the drink gets lower and lower. This causes the backward reaction to take over. Let’s look at that chemical equation again.Īs CO 2 bubbles away from the liquid, the reactants and products move again towards equilibrium. When pop is left out in the open, CO 2 continually escapes. It’s actually because in a fizzy drink, the dilute carbonic acid creates a slight burning sensation on your tongue. Have you ever noticed that fizzy pop tastes better than flat pop? That has nothing to do with the bubbles. This video takes a closer look at carbonated beverages, from seltzer to sparkling wine, to find out what makes them bubbly-and what makes them go flat ( 4:31 min) Why does fizz taste so good? When a can of pop is sealed, high pressure inside the can forces the chemical reaction to the right (the forward reaction). The carbonic acid can be converted back to water and carbon dioxide. That means it can go forwards and backwards. This symbol shows that the reaction is reversible. Notice the symbol ⇋ in the chemical equation. Note that the reactants, H 2O and CO 2, are on the left. The chemical reaction for this process is: H 2O + CO 2 ⇋ H 2CO 3 When CO 2 dissolves in H 2O, water and gaseous carbon dioxide react to form a dilute solution of carbonic acid (H 2CO 3). What chemical reactions are going on inside a pop can? Some CO 2 sits in gas form between the top of the bottle or can and the liquid. Inside the can, CO 2 exists in two forms. This way, the inside of the can maintains enough pressure to prevent the extra CO 2 molecules from escaping. The can is then sealed so that it is airtight. Under these conditions, water molecules can trap lots of CO 2 molecules. To make this happen, manufacturers need to increase the pressure in the can (or bottle) and keep it at a low temperature. Carbonation occurs when carbon dioxide (CO 2) dissolves in water (H 2O) or an aqueous (watery) solution.Ĭarbon dioxide doesn’t easily dissolve in water under everyday conditions. These bubbles happen because of carbonation. Many people love the feeling of bubbles bursting in their mouths when they drink pop. 
Back then, Coca-Cola cost five cents a glass! Why is pop fizzy? It was invented in 1886 by a pharmacist who originally wanted to sell it as a medicine. But did you know that fizz is actually a chemical process? In fact, inside every can or bottle of pop, there is some pretty cool chemistry going on.Ĭoca-Cola is the world’s best-selling pop. Maybe it’s the sight or sound of bubbles rising and popping. When you first open a can of Coca-Cola, what’s the first thing you notice? Maybe it’s that fizzing sound. Open Professional Learning × Close Professional Learning Open Educational Resources × Close Educational Resources 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
